Abstract
Review Article
The Trans-zoonotic Virome interface: Measures to balance, control and treat epidemics
Vinod Nikhra*
Published: 09 April, 2020 | Volume 4 - Issue 1 | Pages: 020-027
The global virome: The viruses have a global distribution, phylogenetic diversity and host specificity. They are obligate intracellular parasites with single- or double-stranded DNA or RNA genomes, and afflict bacteria, plants, animals and human population. The viral infection begins when surface proteins bind to receptor proteins on the host cell surface, followed by internalisation, replication and lysis. Further, trans-species interactions of viruses with bacteria, small eukaryotes and host are associated with various zoonotic viral diseases and disease progression.
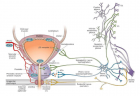
Virome interface and transmission: The cross-species transmission from their natural reservoir, usually mammalian or avian, hosts to infect human-being is a rare probability, but occurs leading to the zoonotic human viral infection. The factors like increased human settlements and encroachments, expanded travel and trade networks, altered wildlife and livestock practices, modernised and mass-farming practices, compromised ecosystems and habitat destruction, and global climate change have impact on the interactions between virome and its hosts and other species and act as drivers of trans-species viral spill-over and human transmission.
Zoonotic viral diseases and epidemics: The zoonotic viruses have caused various deadly pandemics in human history. They can be further characterized as either newly emerging or re-emerging infectious diseases, caused by pathogens that historically have infected the same host species, but continue to appear in new locations or in drug-resistant forms, or reappear after apparent control or elimination. The prevalence of zoonoses underlines importance of the animal–human–ecosystem interface in disease transmission. The present COVID-19 infection has certain distinct features which suppress the host immune response and promote the disease potential.
Treatment for epidemics like covid-19: It appears that certain nutraceuticals may provide relief in clinical symptoms to patients infected with encapsulated RNA viruses such as influenza and coronavirus. These nutraceuticals appear to reduce the inflammation in the lungs and help to boost type 1 interferon response to these viral infections. The human intestinal microbiota acting in tandem with the host’s defence and immune system, is vital for homeostasis and preservation of health. The integrity and balanced activity of the gut microbes is responsible for the protection from disease states including viral infections. Certain probiotics may help in improving the sensitivity and effectivity of immune system against viral infections. Currently, antiviral therapy is available only for a limited number of zoonotic viral infections. Because viruses are intracellular parasites, antiviral drugs are not able to deactivate or destroy the virus but can reduce the viral load by inhibiting replication and facilitating the host’s innate immune mechanisms to neutralize the virus.
Conclusion: Lessons from recent viral epidemics - Considering that certain nutraceuticals have demonstrated antiviral effects in both clinical and animal studies, further studies are required to establish their therapeutic efficacy. The components of nutraceuticals such as luteolin, apigenin, quercetin and chlorogenic acid may be useful for developing a combo-therapy. The use of probiotics to enhance immunity and immune response against viral infections is a novel possibility. The available antiviral therapy is inefficient in deactivating or destroying the infecting viruses, may help in reducing the viral load by inhibiting replication. The novel efficient antiviral agents are being explored.
Read Full Article HTML DOI: 10.29328/journal.abse.1001009 Cite this Article Read Full Article PDF
Keywords:
Virome interface; Zoonotic viral transmission; Viral epidemics; COVID-19; MERS; SARS; Nutraceuticals; Probiotics; Anti-viral agents
References
- Paez-Espino D, Eloe-Fadrosh EA, Pavlopoulos GA, Thomas AD, Huntemann M, et al. Uncovering Earth's virome. Nature. 2016; 536: 425–430. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/27533034
- Edwards RA, Rohwer F. Viral metagenomics. Nature Rev Microbiol. 2005; 3: 504-510. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15886693
- Colson P, Richet H, Desnues C, Balique F, Moal V, et al. Pepper Mild Mottle Virus, a Plant Virus Associated with Specific Immune Responses, Fever, Abdominal Pains, and Pruritus in Humans. PLoS ONE. 2010; 5: e10041. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/20386604
- Zhang T, Breitbart M, Lee WH, Run JQ, Wei CL, et al. RNA viral community in human faeces: prevalence of plant pathogenic viruses. PLoS Biol. 2006; 4: e3. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/16336043
- Mackenzie JS, Jeggo M. Reservoirs and vectors of emerging viruses. Curr Opin Virol. 2013; 3: 170-179. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/23491947
- Carrol D, Watson B, Togami E, Daszak P, Mazet JA, et al. Building a global atlas of zoonotic viruses. Bull World Health. 2018; 96: 292–294. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/29695886
- Olival KJ, Hosseini PR, Zambrana-Torrelio C, Ross N, Bogich TL, et al. Host and viral traits predict zoonotic spill-over from mammals. Nature. 2017; 546: 646-650. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/28636590
- Morse SS, Mazet JA, Woolhouse M, Parrish CR, Carroll D, et al. Prediction and prevention of the next pandemic zoonosis. Lancet. 2012; 380: 1956-1965. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/23200504
- Lozano R, Naghavi M, Foreman K, Lim S, Shibuya K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012; 380: 2095–2128. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/23245604
- Jones KE, Patel NG, Levy MA, Storeygard A, Balk D, et al. Global trends in emerging infectious diseases. Nature. 2008; 451: 990-993. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/18288193
- Fauci AS, Morens DM. The perpetual challenge of infectious diseases. N Engl J Med. 2012; 366: 454–461. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/22296079
- Bolles M, Donaldson E, Baric R. SARS-CoV and emergent coronaviruses: Viral determinants of interspecies transmission. Curr Opin Virol. 2011; 1: 624–634. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/22180768
- Graham RL, Baric RS. Recombination, reservoirs, and the modular spike: Mechanisms of coronavirus cross-species transmission. J Virol. 2010; 84: 3134–3146. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/19906932
- Graham RL, Donaldson EF, Baric RS. A decade after SARS: Strategies for controlling emerging coronaviruses. Nature Rev Microbiol. 2013; 11: 836-848. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/24217413
- Li F. Receptor recognition and cross-species infections of SARS coronavirus. Antiviral Res. 2013; 100: 246–254. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/23994189
- Johnson CK, Hitchens PL, Evans TS, Goldstein T, Thomas K, et al. Spillover and pandemic properties of zoonotic viruses with high host plasticity. Sci Rep. 2015; 5: 14830. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26445169
- McCarty MF, DiNicolantonio JJ. Nutraceuticals have potential for boosting the type 1 interferon response to RNA viruses including influenza and coronavirus. Prog Cardiovasc Dis. 2020. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/32061635
- Brayden Humpherys B, Busath DD. Anti-Influenza Nutraceuticals: Antiviral and Anti-Inflammatory Effects. Advances in Complementary & Alternative medicine. 2019; 4.
- To EE, Luong R, Diao J, O' Leary JJ, Brooks DA, et al. Novel endosomal NOX2 oxidase inhibitor ameliorates pandemic influenza A virus‐induced lung inflammation in mice. Respirology. 2019; 24: 1011–1017. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/30884042
- Vetvicka V, Vetvickova J. Glucan supplementation enhances the immune response against an influenza challenge in mice. Ann Transl Med. 2015; 3: 22. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/25738142
- Hawkins J, Baker C, Cherry L, Dunne E. Black elderberry (Sambucus nigra) supplementation effectively treats upper respiratory symptoms: a meta-analysis of randomized, controlled clinical trials. Complement Ther Med. 2019; 42; 361-365. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/30670267
- Li N, Ma W-T, Pang M, Fan QL, Hua JL. The Commensal Microbiota and Viral Infection: A Comprehensive Review. Front Immunol. 2019; 10: 1551.PubMed: https://www.ncbi.nlm.nih.gov/pubmed/31333675 .
- Lehtoranta L, Pitkäranta A, Korpela R. Probiotics in respiratory virus infections. Eur J Clin Microbiol Infect Dis. 2014; 33: 1289-1302. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/24638909
- O’Toole PW, Marchesi JR, Hill C. Next-generation probiotics: the spectrum from probiotics to live biotherapeutics. Nat Microbiol. 2017; 2: 17057. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/28440276
- Maeda N, Nakamura R, Hirose Y, Murosaki S, Yamamoto Y, et al. Oral administration of heat-killed lactobacillus plantarum l-137 enhances protection against influenza virus infection by stimulation of type I interferon production in mice. Int Immunopharmacol. 2009; 9; 11-22. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/19410659
- Yang Y, Song H, Wang L, Palissa C, Esch B, et al. Antiviral Effects of a Probiotic Metabolic Products against Transmissible Gastroenteritis Coronavirus. Arch Virol. 2013; 158: 799-807. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/23188495
- Harata G, He F, Hiruta N, Kawase M, Kubota A, et al. Intranasal administration of Lactobacillus rhamnosus GG protects mice from H1N1 influenza virus infection by regulating respiratory immune responses. Lett Appl Microbiol, 2010; 50; 597–602. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/20438620
- Horie A, Tomita Y, Ohshio K, Fujiwara D, Fujii T. Characterization of genomic DNA of lactic acid bacteria for activation of plasmacytoid dendritic cells. BMC Microbiol. 2019; 19: 88. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/31060586
- Kumar R, Seo BJ, Mun MR, Kim CJ, Lee I, et al. Putative probiotic lactobacillus spp. from porcine gastrointestinal tract inhibit transmissible gastroenteritis coronavirus and enteric bacterial pathogens. Trop Anim Health Prod. 2010; 42: 1855-1860. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/20623187
- Chai W, Burwinkel M, Wang Z, Palissa C, Esch B, et al. Antiviral effects of a probiotic enterococcus faecium, strain against transmissible gastroenteritis coronavirus. Arch\Virol. 2013; 158: 799-807. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/23188495
- Lu W, Feng Y, Jing F, Han Y, Lyu N, et al. Association between gut microbiota and CD4 recovery in HIV-1 infected patients. Front Microbiol. 2018; 9: 1451. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/30034377
- Razonable RR. Antiviral Drugs for Viruses Other Than Human Immunodeficiency Virus. Mayo Clin Proc. 2011; 86: 1009–1026. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/21964179
- Lin GL, McGinley JP, Drysdale SB, Pollard AJ. Epidemiology and Immune Pathogenesis of Viral Sepsis. Front Immunol. 2018; 9: 2147. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/30319615
- Influenza (Flu) Antiviral Drugs and Related Information.
Figures:
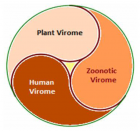
Figure 1
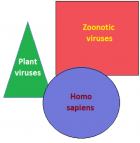
Figure 2
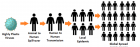
Figure 3
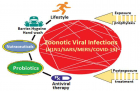
Figure 4

Figure 5
Similar Articles
-
The Trans-zoonotic Virome interface: Measures to balance, control and treat epidemicsVinod Nikhra*. The Trans-zoonotic Virome interface: Measures to balance, control and treat epidemics. . 2020 doi: 10.29328/journal.abse.1001009; 4: 020-027
-
Applications of convolutional neural networks in chest X-ray analyses for the detection of COVID-19Patrick Ting*,Anish Kasam,Kevin Lan. Applications of convolutional neural networks in chest X-ray analyses for the detection of COVID-19. . 2022 doi: 10.29328/journal.abse.1001015; 6: 001-007
-
Using Model Classification to detect Bias in Hospital TriagingPatrick Ting*, Aayaan Sahu, Nishad Wajge, Vineet Rao, Hiresh Poosarla, Phil Mui. Using Model Classification to detect Bias in Hospital Triaging. . 2023 doi: 10.29328/journal.abse.1001022; 7: 024-030
Recently Viewed
-
Addiction to self-strangulation: a case-reportAurely Ameller*,Yann Le Strat,Marion Cadranel,Celine Portalier, Caroline Dubertret. Addiction to self-strangulation: a case-report . J Addict Ther Res. 2017: doi: 10.29328/journal.jatr.1001003; 1: 016-021
-
Nanoencapsulated Extracts from Leaves of Bauhinia forficata Link: In vitro Antioxidant, Toxicogenetic, and Hypoglycemic Activity Effects in Streptozotocin-induced Diabetic MiceBárbara Verônica Cardoso de Souza, Alessandra Braga Ribeiro*, Rita de Cássia Meneses Oliveira, Julianne Viana Freire Portela, Ana Amélia de Carvalho Melo Cavalcante, Esmeralda Maria Lustosa Barros, Luís Felipe Lima Matos, Tarsia Giabardo Alves, Maria. Nanoencapsulated Extracts from Leaves of Bauhinia forficata Link: In vitro Antioxidant, Toxicogenetic, and Hypoglycemic Activity Effects in Streptozotocin-induced Diabetic Mice. Arch Pharm Pharma Sci. 2024: doi: 10.29328/journal.apps.1001063; 8: 100-115
-
Oral Suspension as Versatile Galenic Formulation in PediatryMauro Luisetto*, Almukthar N, Edbey K, Mashori GR, Fiazza C, Dona’ l, Cabianca L, Latyshev O. Oral Suspension as Versatile Galenic Formulation in Pediatry. Arch Pharm Pharma Sci. 2024: doi: 10.29328/journal.apps.1001062; 8: 091-099
-
Modulation of Microbiota and its Impact on DepressionKousik Maparu*. Modulation of Microbiota and its Impact on Depression. Arch Pharm Pharma Sci. 2024: doi: 10.29328/journal.apps.1001061; 8: 089-090
-
Experiences of Consumers on the Health Effects of Fake and Adulterated Medicines in NigeriaChijioke M Ofomata, Nkiru N Ezeama, Chinelo Ezejiegu*. Experiences of Consumers on the Health Effects of Fake and Adulterated Medicines in Nigeria. Arch Pharm Pharma Sci. 2024: doi: 10.29328/journal.apps.1001059; 8: 075-081
Most Viewed
-
Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth EnhancersH Pérez-Aguilar*, M Lacruz-Asaro, F Arán-Ais. Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth Enhancers. J Plant Sci Phytopathol. 2023 doi: 10.29328/journal.jpsp.1001104; 7: 042-047
-
Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case PresentationJulian A Purrinos*, Ramzi Younis. Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case Presentation. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001099; 8: 075-077
-
Feasibility study of magnetic sensing for detecting single-neuron action potentialsDenis Tonini,Kai Wu,Renata Saha,Jian-Ping Wang*. Feasibility study of magnetic sensing for detecting single-neuron action potentials. Ann Biomed Sci Eng. 2022 doi: 10.29328/journal.abse.1001018; 6: 019-029
-
Pediatric Dysgerminoma: Unveiling a Rare Ovarian TumorFaten Limaiem*, Khalil Saffar, Ahmed Halouani. Pediatric Dysgerminoma: Unveiling a Rare Ovarian Tumor. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001087; 8: 010-013
-
Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative reviewKhashayar Maroufi*. Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative review. J Sports Med Ther. 2021 doi: 10.29328/journal.jsmt.1001051; 6: 001-007

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."